Use of camera traps to investigate if the presence of invasive Japanese knotweed (Fallopia japonica) affects wildlife abundance along the Bronx River, New York City, USA.
Baja 1,2016 Miami University- Global Field Program
Michelle Medina Senior Wild Animal Keeper
Bronx Zoo/Wildlife Conservation Society
Bronx, New York, USA
Abstract
The highly invasive plant, Japanese knotweed (Fallopia japonica) has been found to diminish invertebrate and aquatic communities. Researchers hypothesize Japanese knotweed could overtime negatively affect amphibians, reptiles, mammals and birds. Camera traps were utilized to investigate if the presence of Japanese knotweed negatively affects wildlife along the Bronx River, New York City, NY, USA. Camera traps were installed at two sites with similar habitat characteristics, the exception that one site was near a stand of Japanese knotweed while the other with only native vegetation. The prediction, if Japanese knotweed negatively affects wildlife abundance, there would be fewer images of wildlife recorded at the site with the invasive. Identical cameras were deployed simultaneously, model Browning BTC-5HD, for 42 days. Mammals and birds were detected at both sites. Species richness, relative abundance and daily camera encounter rate data were collected from the recorded images. While the invasive site’s camera recorded a higher number of encounters with mammals, the native site’s camera recorded a higher number of encounters with birds. The invasive site showed a decrease in biodiversity, upon comparison of both Shannon Wiener and Simpson indices, in both mammals and birds, as well as a decline in bird species richness.
Keywords: camera trap, Japanese knotweed, wildlife abundance, biodiversity, invasive species.
Keywords: camera trap, Japanese knotweed, wildlife abundance, biodiversity, invasive species.
Use of camera traps to investigate if the presence of invasive Japanese knotweed (Fallopia japonica) affects wildlife abundance along the Bronx River, New York City, USA.
Introduction
Japanese knotweed (Fallopia japonica) is one of the most prolific invasive species of vegetation that has overrun riparian corridors in eastern and northwest North America as well as parts of Europe, and the United Kingdom (Gerber, et al., 2008; Urgenson et al., 2009; USDA, 2016). Japanese knotweed was originally imported into the United States and Europe as ornamental plants from Asia in the late 1800s. Though Japanese knotweed is pollinated by insects and its seeds dispersed by wind, in riparian areas the principal method of reproduction is via growth of its large rhizomes (USDA, 2016). The plant disperses via small pieces of rhizome that easily break off during flooding and carried downstream, which grow into large plants quickly (Urgenson et al., 2009; USDA, 2016). Japanese knotweed grows in large dense stands, like bamboo, reaching heights of 5m, with rhizomes which can extend 2m into the ground, and its biomass can surpass 200 tons/ha (Urgenson et al., 2009; USDA, 2016).
Japanese knotweed stands have been found to dominate riparian habitats as well as disturbed areas, including roadsides (Urgenson et al., 2009; Weston, Barney, & DiTommaso, 2005). Due to their large rhizome roots, when Japanese knotweed invades river and stream banks it causes them to be less stable, compared to fine rooted native vegetation (Gerber et al., 2008). This leads to erosion of banks and diminishes protection for terrestrial invertebrate and aquatic species (Gerber et al., 2008).
Studies of Japanese knotweed have shown it to not only displace native vegetation, but to also limit the light reaching the soil and shorter vegetation; its dominance decreases the biodiversity of native vegetation as well as terrestrial invertebrates (Gerber et al., 2008; Urgenson et al., 2009). For example, Gerber et al. (2008) found the amount of biomass of invertebrates in areas dominated by knotweeds was half that measured in habitats dominated by bush and grasslands. The proliferation and dominance of Japanese knotweed and the associated decrease of invertebrate biomass, which acts as a food source, has led researchers to believe that long-term changes in riparian habitats are likely to have trophic consequences affecting amphibians, reptiles, birds, and mammals (Gerber et al., 2008; Maerz et al., 2005). Maerz et al. (2005) have shown that in habitats dominated by Japanese knotweed in Broome County, NY, USA, green frogs (Rana clamitans) demonstrated decreased foraging success. The researchers compared the body mass of frogs foraging in sites invaded by Japanese knotweed versus frogs foraging in areas of native vegetation, and concluded those foraging in native areas gained more body mass than those foraging in the knotweed areas (Maerz et al., 2005).
Japanese knotweed is difficult to remove once it has taken hold in an area. Since Japanese knotweed can grow quickly from small pieces, cutting and completely removing all remaining pieces is labor intensive and can take years (Weston et al., 2005; Haight et al., 2014). While some herbicides have been found to be effective on knotweed, many are not suggested for use in areas along rivers and streams, due to their non-selectiveness and risk to aquatic communities (Weston, Barney, & DiTommaso, 2005). Haight et al. (2014) compared the effects of different removal techniques along the portion of the Bronx River in the New York Botanical Garden, Bronx, NY (located north of the Bronx Zoo). Haight et al. (2014) concluded that even after removal, Japanese knotweed returns and reestablishes itself quickly. However, they found re-growth might be suppressed by planting native vegetation, especially oak-saplings, when combined with constant removal of the knotweed (Haight et al., 2014).
The Bronx River is New York City’s (NYC) only freshwater river, and flows 23 miles south from Westchester County through The Bronx, and ending in the East River and Long Island Sound (Haight et al., 2014). The Bronx River forms a corridor and is habitat for urban wildlife. Large stands of Japanese knotweed have been observed along the portion of the Bronx River, which flows through the Bronx Zoo (Medina, 2014).
With researchers demonstrating trophic effects due to Japanese knotweed (Gerber et al., 2008; Maerz et al., 2005) I chose to investigate if Japanese knotweed affects the abundance of wildlife along the Bronx River. Camera traps were deployed to compare the relative abundances of mammals and birds at a site adjacent a large stand of Japanese knotweed versus a site without Japanese knotweed. The assumption, supported by research, that the number of images captured by the cameras per unit time, the ‘trapping rate’, is related to the density of a species (Rowcliffe, Turvey, & Carbone, 2008). If the Japanese knotweed negatively affects the occurrence and abundance of wildlife, then I would predict there would be a lower rate of encounters detected by the cameras at the invasive site, with Japanese knotweed, versus the native site.
Camera traps have been used for fifty years to record wildlife activity (Swann, Hass, Dalton, & Wolf, 2004). Infrared-triggered cameras have increasingly been used in wildlife studies, for inventory, behavioral studies, population estimates, relative abundance, and habitat use (Swann et al., 2004). An infrared beam emitted by the camera detects motion when an animal enters the detection zone, and triggers the camera to capture an image (Karlin & DeLaPaz, 2015). Camera traps minimize human disturbance and are especially helpful for studying cryptic species, in regions that are difficult to navigate, and for those that encounter harsh weather conditions (Trolliet et al., 2014).
Japanese knotweed stands have been found to dominate riparian habitats as well as disturbed areas, including roadsides (Urgenson et al., 2009; Weston, Barney, & DiTommaso, 2005). Due to their large rhizome roots, when Japanese knotweed invades river and stream banks it causes them to be less stable, compared to fine rooted native vegetation (Gerber et al., 2008). This leads to erosion of banks and diminishes protection for terrestrial invertebrate and aquatic species (Gerber et al., 2008).
Studies of Japanese knotweed have shown it to not only displace native vegetation, but to also limit the light reaching the soil and shorter vegetation; its dominance decreases the biodiversity of native vegetation as well as terrestrial invertebrates (Gerber et al., 2008; Urgenson et al., 2009). For example, Gerber et al. (2008) found the amount of biomass of invertebrates in areas dominated by knotweeds was half that measured in habitats dominated by bush and grasslands. The proliferation and dominance of Japanese knotweed and the associated decrease of invertebrate biomass, which acts as a food source, has led researchers to believe that long-term changes in riparian habitats are likely to have trophic consequences affecting amphibians, reptiles, birds, and mammals (Gerber et al., 2008; Maerz et al., 2005). Maerz et al. (2005) have shown that in habitats dominated by Japanese knotweed in Broome County, NY, USA, green frogs (Rana clamitans) demonstrated decreased foraging success. The researchers compared the body mass of frogs foraging in sites invaded by Japanese knotweed versus frogs foraging in areas of native vegetation, and concluded those foraging in native areas gained more body mass than those foraging in the knotweed areas (Maerz et al., 2005).
Japanese knotweed is difficult to remove once it has taken hold in an area. Since Japanese knotweed can grow quickly from small pieces, cutting and completely removing all remaining pieces is labor intensive and can take years (Weston et al., 2005; Haight et al., 2014). While some herbicides have been found to be effective on knotweed, many are not suggested for use in areas along rivers and streams, due to their non-selectiveness and risk to aquatic communities (Weston, Barney, & DiTommaso, 2005). Haight et al. (2014) compared the effects of different removal techniques along the portion of the Bronx River in the New York Botanical Garden, Bronx, NY (located north of the Bronx Zoo). Haight et al. (2014) concluded that even after removal, Japanese knotweed returns and reestablishes itself quickly. However, they found re-growth might be suppressed by planting native vegetation, especially oak-saplings, when combined with constant removal of the knotweed (Haight et al., 2014).
The Bronx River is New York City’s (NYC) only freshwater river, and flows 23 miles south from Westchester County through The Bronx, and ending in the East River and Long Island Sound (Haight et al., 2014). The Bronx River forms a corridor and is habitat for urban wildlife. Large stands of Japanese knotweed have been observed along the portion of the Bronx River, which flows through the Bronx Zoo (Medina, 2014).
With researchers demonstrating trophic effects due to Japanese knotweed (Gerber et al., 2008; Maerz et al., 2005) I chose to investigate if Japanese knotweed affects the abundance of wildlife along the Bronx River. Camera traps were deployed to compare the relative abundances of mammals and birds at a site adjacent a large stand of Japanese knotweed versus a site without Japanese knotweed. The assumption, supported by research, that the number of images captured by the cameras per unit time, the ‘trapping rate’, is related to the density of a species (Rowcliffe, Turvey, & Carbone, 2008). If the Japanese knotweed negatively affects the occurrence and abundance of wildlife, then I would predict there would be a lower rate of encounters detected by the cameras at the invasive site, with Japanese knotweed, versus the native site.
Camera traps have been used for fifty years to record wildlife activity (Swann, Hass, Dalton, & Wolf, 2004). Infrared-triggered cameras have increasingly been used in wildlife studies, for inventory, behavioral studies, population estimates, relative abundance, and habitat use (Swann et al., 2004). An infrared beam emitted by the camera detects motion when an animal enters the detection zone, and triggers the camera to capture an image (Karlin & DeLaPaz, 2015). Camera traps minimize human disturbance and are especially helpful for studying cryptic species, in regions that are difficult to navigate, and for those that encounter harsh weather conditions (Trolliet et al., 2014).
Methods
Study area
The study area was located along the Bronx River, Bronx, NY, USA. Two sites were chosen for camera installation along the eastern bank of the Bronx River within the Bronx Zoo (see Map 1). The area is not accessible by zoo visitors, the only public access being by infrequent paddlers using the river. The two sites were chosen as they offered similar wildlife habitat features, with the notable exception, that site #1 had no Japanese knotweed immediately present, and site #2 was located near a large stand of Japanese knotweed.
The study area was located along the Bronx River, Bronx, NY, USA. Two sites were chosen for camera installation along the eastern bank of the Bronx River within the Bronx Zoo (see Map 1). The area is not accessible by zoo visitors, the only public access being by infrequent paddlers using the river. The two sites were chosen as they offered similar wildlife habitat features, with the notable exception, that site #1 had no Japanese knotweed immediately present, and site #2 was located near a large stand of Japanese knotweed.
Native site. Site #1 was located at N40°50’47” W 73°52’28”. No Japanese knotweed was present within 45m of Site #1. The camera was installed 0.5m above the ground on a Red maple tree, and orientated toward N 344°W. The camera was located 7.6m from the river’s edge. There was a live Northern red oak tree which had fallen in the river, with half of its trunk on land, located 5.2m in front of the camera (see Map 2). The immediate area in front of the camera was relatively clear of vegetation, which would help diminish false detections triggered by vegetation moving in wind (Swann et al., 2004). The surrounding area had many beech trees.
Invasive site. Site #2 was located 111m north of site#1, at N40°50’50” W73°52’27”. The camera was attached 0.4m above the ground on a Red maple tree. The camera was oriented N20°E, and located 1m from the river’s edge. Similar to site #1, there was a fallen tree (live Red maple), located 5m from the camera. A large stand of Japanese knotweed was located 6m in front of the camera, behind the fallen tree (see Map 3). The Japanese knotweed stand encompassed an area 15m in length along the river’s edge, and 5m up the bank, roughly 75m². The trees in the adjacent area to the camera were beech and oak. A small branch was removed from the immediate foreground of the camera to diminish false positive detections triggered by movement of the branch in the wind (Swann et al., 2004).
Camera trapping
Two infrared-triggered trail cameras were simultaneously deployed, one at each site. Both cameras were the same model for consistency, Browning model BTC-5HD (Prometheus Group, LLC, Birmingham, Alabama, USA). The photo options were set the same for both cameras, operational mode: trail cam, which sets for still photographs; photo quality was 10MP for each image; the picture delay was set to one minute and multi-shot mode was selected to take a series of eight rapid images 0.3 seconds apart. Therefore, eight photographs were captured for every event detected; the camera would then delay one minute before another detection event could trigger the camera. Neither camera site was baited.
The field of view of this camera model is 55 degrees wide. The infrared detection zone is on average 16.7m, and the infrared flash illuminates 30.5m. Each camera was tightly mounted to a tree, using a nylon strap and secured with a locked cable. It is important that the camera be tightly mounted to a rigid structure to minimize any movement of the camera sensor, which could falsely trigger the camera (Swann et al., 2004). The images were recorded on an SD card and each camera used six AA batteries. Each image is stamped with the date and time, temperature, moon phase and site name. The camera trapping session lasted 42 days, first deployed on September 13, 2016 at ~ 3:15pm (site#1) and ~3:40pm (site#2), and retrieved on October 22, 2016 at 3:20pm (site#2) and 3:47pm (site#1). The cameras were checked three times during their deployment, for proper placement, battery operation and to change SD cards.
Data analysis
Because species detected by the cameras were not easily individually identifiable, a 30-minute rule was used to define an individual encounter or sighting (Burton et al., 2015; Christensen, 2016; Karlin & De La Paz, 2015). For example, if there was an image recorded of a chipmunk at 8:00am, and more images recorded at the same site of a chipmunk at 8:07am, 8:12am, and 8:22am, I recorded the images as one chipmunk in my data, and did not count another chipmunk until after 8:30am. The exception to this rule being those times when an individual displayed a visual means or characteristic to identify it as a new separate individual e.g. gray versus black color morphs of the Gray squirrel (Sciurus carolinensis), juvenile versus adult raccoons, or multiple individuals.
Data from each image (species, number of individuals, visual characteristics) was logged by date and time. The daily encounters (independent sightings) at each site were tallied over the camera-trapping period. The camera trap encounter rates of each of the species detected at the two sites were calculated (Bowkett, Rovero & Marshall, 2007; Christensen, 2016).
Camera encounter rate = Total # independent sightings of observed species from camera
Total # of camera days for study period
Using this data, the Student’s t-test was used, with α=0.05, to determine if there was a difference in numbers of independent sightings of all species detected at the two sites. The Shannon-Wiener diversity index (H), and Simpson diversity index (D) was calculated for both mammal and bird species diversity using species richness and abundance data collected from the images.
Results
The two cameras were simultaneously deployed and functioned for 42 consecutive days. They detected 1316 events in total, since I set the cameras to capture eight images per detection event, this resulted in 10,528 image files collected to be processed. The native site detected 601 events, with 4,808 image files captured. From these, using the ‘30-minute rule’, 351 wildlife triggered events were defined as individual encounters at the native site. The invasive site detected 715 total events, capturing 5,720 image files. Of these events, 414 were defined as individual encounters, using the ’30-minute’ rule. On two occasions zookeepers searching for browse, i.e. edible vegetation for collection animals, triggered a camera. There were few false positives, usually triggered by wind, identified by vegetation moving upon viewing the series of images. Both cameras did not detect any events during the daytime of October 21, 2016. There was torrential rainfall on this day, resulting in the flooding of the detection zone of site #1, which lasted at least 7.5 hours. Images were recorded that afternoon of wood ducks (Aix sponsa) swimming, and that night of raccoons foraging in the floodwaters (Procyon lotor). The highest temperature recorded during deployment by the cameras was 32°C, with the lowest 3°C.
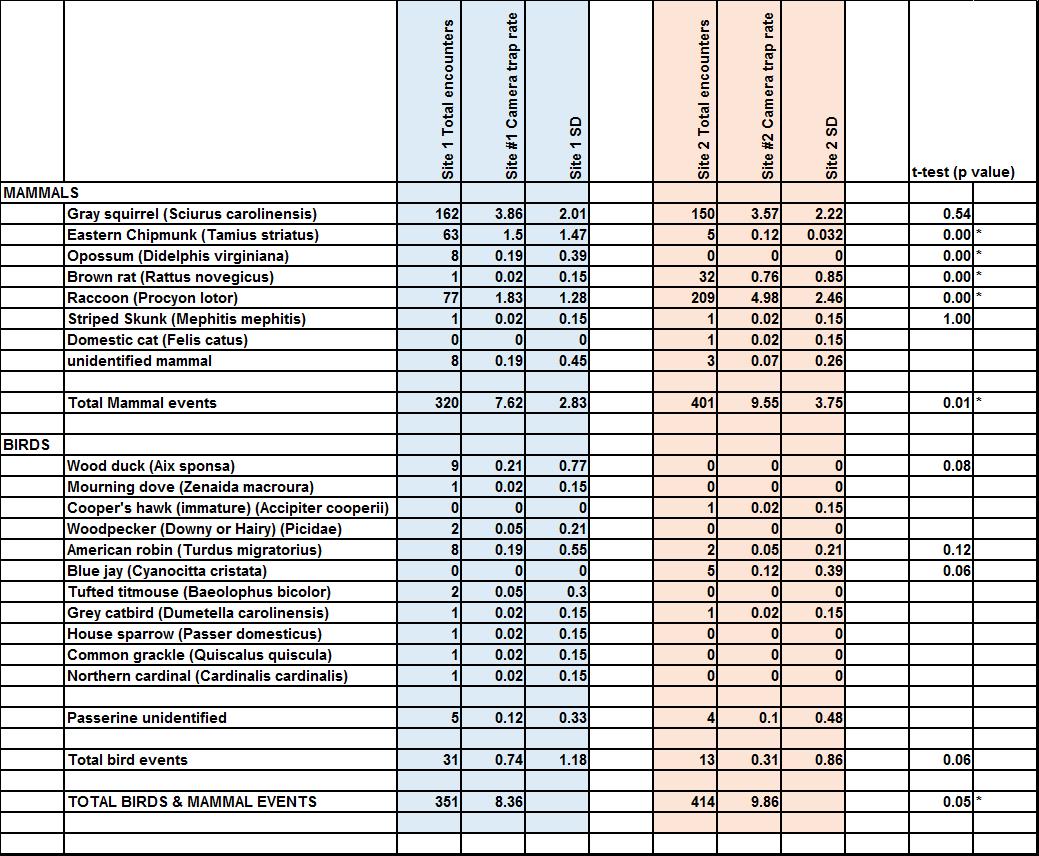
Mammals and birds triggered the camera traps at both sites. The Student’s t-test, α=0.05, revealed a nearly significant difference of total bird and mammal encounters at the sites (p=0.051) (Table 1). The Student’s t-test, α=0.05, revealed there was a difference in the camera encounter trap rate of mammals between the two sites, with a higher number of mammal encounters occurring at the invasive site. The most common animals detected by the cameras at both sites were Gray squirrels and raccoons (Table 1). A high number of Eastern chipmunk (Tamius striatus) encounters, 63, were recorded at the native site, with only five encounters recorded at the invasive site.
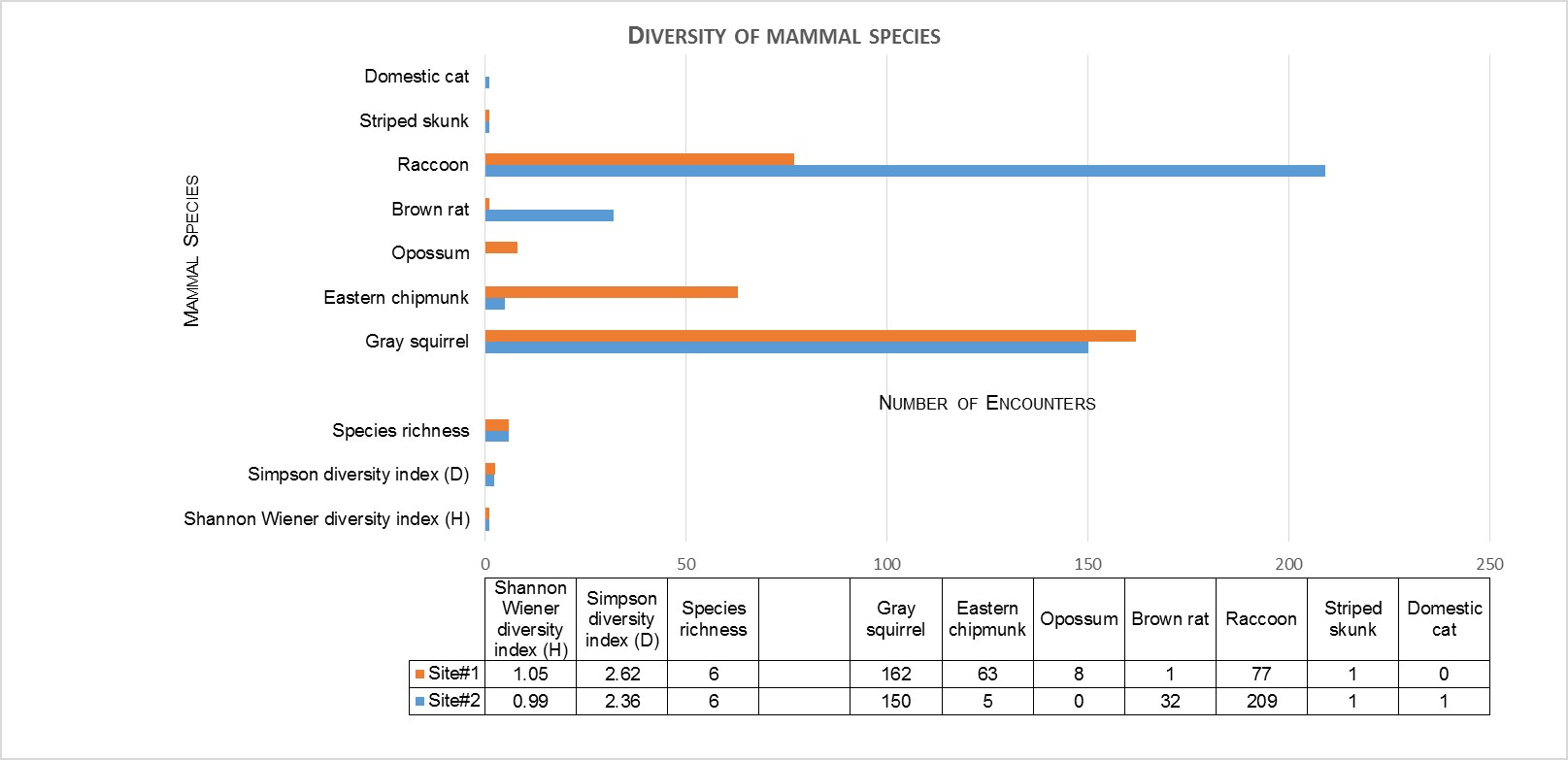
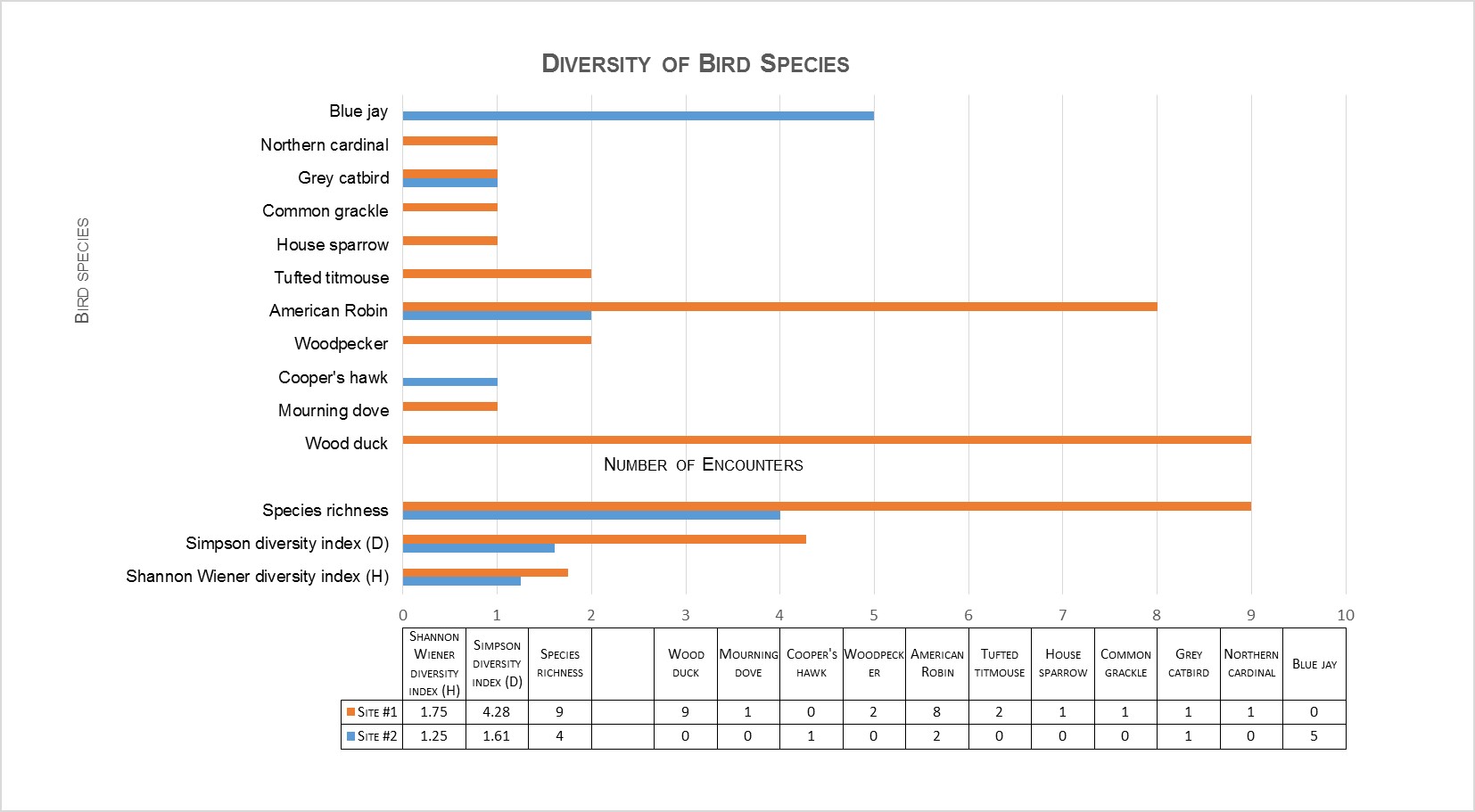
When comparing the camera trap encounter rates for each mammal species at their respective sites, Student’s t-tests revealed there were differences among the two sites for Eastern chipmunk and opossum (Didelphis virginiana), with these two species occurring most often at the native site. The Student’s t-test also revealed differences for brown rat (Rattus norvegicus), and raccoon (Procyon lotor), with these two species occurring more often at the invasive site. Both the Shannon’s diversity index (H), and Simpson’s diversity index (D) showed a decrease in both mammal (Figure 1) and bird diversity at the invasive site (Figure 2).
Mammals and birds triggered the camera traps at both sites. The Student’s t-test, α=0.05, revealed a nearly significant difference of total bird and mammal encounters at the sites (p=0.051) (Table 1). The Student’s t-test, α=0.05, revealed there was a difference in the camera encounter trap rate of mammals between the two sites, with a higher number of mammal encounters occurring at the invasive site. The most common animals detected by the cameras at both sites were Gray squirrels and raccoons (Table 1). A high number of Eastern chipmunk (Tamius striatus) encounters, 63, were recorded at the native site, with only five encounters recorded at the invasive site.
When comparing the camera trap encounter rates for each mammal species at their respective sites, Student’s t-tests revealed there were differences among the two sites for Eastern chipmunk and opossum (Didelphis virginiana), with these two species occurring most often at the native site. The Student’s t-test also revealed differences for brown rat (Rattus norvegicus), and raccoon (Procyon lotor), with these two species occurring more often at the invasive site. Both the Shannon’s diversity index (H), and Simpson’s diversity index (D) showed a decrease in both mammal (Figure 1) and bird diversity at the invasive site (Figure 2).
Table 1 Compiled event data from site #1 & site #2; Student's t-test p-values for mean daily events.
|
An “unidentified mammal” was defined as unknown animal under or too close to camera identify but fur, hair, whiskers were visible. “Passerine unidentified” was defined as a perching bird which was not identifiable due to image quality or obscured view.
|
|
Figure 1 Diversity of mammal species at both sites: Shannon and Simpson diversity indices
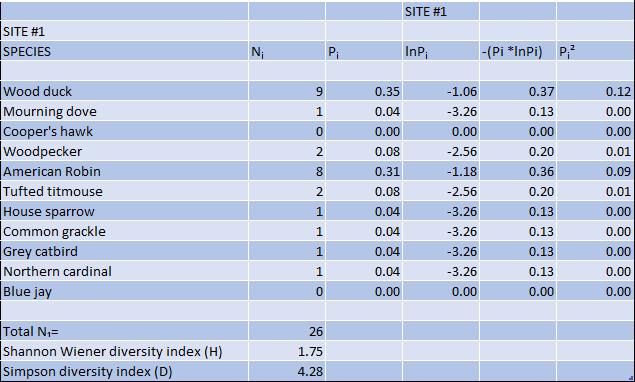
Figure 2 Diversity of bird species at both sites: Shannon and Simpson diversity indices
Discussion
Camera trapping
Camera traps have been used for 50 years to study wildlife; some uses include inventory, population estimates, animal densities, relative abundance, habitat use and behavioral studies (Bowkett et al., 2007; Rowcliffe et al., 2008; Swann et al., 2004; Trolliet et al., 2014). Less expensive, commercially available infrared units targeted for recreational hunters have flooded the market further increasing their use among wildlife managers and conservationists (Newey et al., 2015). Two such trail cameras were deployed for this project to investigate if there was an effect of Japanese knotweed on the occurrence and abundance of wildlife. Large stands of Japanese knotweed have been observed along the Bronx River, Bronx, NY (Medina, 2014). Japanese knotweed has been shown to have an associated effect of decreasing invertebrate biomass (Gerber et al., 2008). Since researchers hypothesize that long-term changes to riparian habitats due to Japanese knotweed are likely, and could have trophic consequences affecting wildlife (Gerber et al., 2008; Maerz et al., 2005)
I chose to investigate if Japanese knotweed affects the abundance of mammals and birds along the Bronx River, Bronx, NY. If the Japanese knotweed negatively affects the occurrence and relative abundance of wildlife, I predicted there would be a lower rate of wildlife encounters detected by the cameras at the site with Japanese knotweed. The assumption, supported by research, is that numbers of images captured by the cameras per unit time, the ‘trapping rate’, is related to the density, and relative abundance of a species (Christensen, 2016; Rowcliffe et al., 2008).
The two sites were selected due to both containing similar wildlife habitat variables; both were near the river, and by a large, fallen, live tree, which could offer food, protection, or a den site for wildlife. The major difference between the sites was site #2 had a large stand of Japanese knotweed present, and site #1 did not have knotweed immediately present. Two identical cameras were set to function alike (i.e. time, image quality, photo delay, number of photographs to capture for an event). An assumption is the probability of false negatives, and the probability of detecting the same species will be the same for both sites by using the same model camera, with the same settings (Newey et al., 2015; O’Brien, 2010). A false negative occurs when there is an animal present but not detected by a camera, the lack of a detection of a species or individual by a camera is not proof of its absence. The probability of detection is the chance that an individual will be detected and photographed if it is present during sampling (O’Brien, 2011). While I assume the detection probability to be the same for both sites using the same camera model, I do not know what the actual detection probability is. Therefore, since abundance cannot be estimated a relative abundance index was used, the camera encounter rate, which is commonly used when studying animals that are not individually identifiable (Christensen, 2016). A relative abundance index is assumed to be correlated to population size (Christensen, 2016; O’Brien, 2011). The camera encounter rate is the number of detections of a species per unit time (Christensen, 2016). I calculated the daily encounter rates of all species detected at both sites.
By utilizing the eight-shot, multi-shot burst mode on both cameras, I could log animal encounters I may have missed if this setting had been a lower number of photographs per event. Some species are small and move quickly, i.e. Eastern chipmunks, and on several occasions, it was solely the fact it was a series of eight images that the chipmunk or bird that triggered the event could be observed. If the camera had been set to take only one to three photographs per event, it is likely that the individual would have been missed. In addition to differing species characteristics, i.e. size and quickness, there are other factors that can affect the efficiency of camera trapping, including sex, season and camera position (Hamel et al., 2012; Rowcliffe et al., 2008). The cameras were positioned as similar as possible using the available trees at the selected sites, and both cameras were deployed simultaneously so weather conditions, season, and temperature would be the same at both sites during the camera trap session.
An assumption of researchers using camera traps is the animals move independently of the cameras, and there is no “trap shyness” (Rowcliffe et al., 2008). While I may hold to this assumption that the presence of the cameras did not inhibit the movement or presence of wildlife, it was apparent, especially in the first few days and nights that some animals were aware and acknowledged the cameras. Images of raccoons on the first night, recorded behavior suggesting they were curious of the camera, as well as some squirrels on the first few days. The camera traps were deployed for 42 days. Hamel et al. (2003) concluded from their studies that a minimum of 20-30 problem free days of camera trap deployment are required for the stabilization of occupancy and detection probabilities, and for their precision to be maximized.
Mammals
Mammals triggered the cameras at both sites. The higher number of total mammal encounters (raw) occurred at the invasive site, with 401 encounters, compared to 320 encounters at site #1 (Table 1). The Student’s t-test revealed there was a difference, (p=0.01, α=0.05), in the daily camera encounter rates between the two sites. Contrary to my prediction, the data revealed more mammal detections occurred at the site with the Japanese knotweed. I suspect this resulted from a family of raccoons that inhabited a den near the fallen tree at site #2. This circumstance created a source of bias and skewed the abundance for raccoons and therefore total mammals at the invasive site.
While species richness, the number of species represented, was the same for both sites, with six mammal species detected, abundance data was further utilized to measure biodiversity. The Shannon-Wiener and Simpson diversity indices revealed a slightly higher biodiversity occurring at the native site (Figure 1). This measure supports my prediction of Japanese knotweed negatively affecting mammal communities.
Raccoon. Raccoons were detected at both sites. Using the 30-minute rule, there were 77 encounters at the native site, and 209 encounters recorded at the invasive site. The Student’s t-test revealed a statistical difference (p=0.00, α=0.05) in daily encounters not due to chance alone. As previously discussed, I suspect this difference was due to a family of raccoons inhabiting a den located behind the fallen tree at site #2, near the Japanese knotweed. Raccoons were detected most often during nighttime, with only three encounters in mornings; this conforms to the published behavioral studies (Elbroch & Rinehart, 2011).
Eastern chipmunk. Eastern chipmunks were detected at both sites, with a higher number of encounters detected at the site with no Japanese knotweed. The Student’s t-test revealed a statistical difference, (p=0.00, α=0.05) between daily encounters at the two sites. The findings for Eastern chipmunk encounters support the prediction of decreased abundance at the invasive site. Upon retrieving the cameras I observed the camera’s detection zone, at site #1, was littered with acorns, an abundant food source for chipmunks and squirrels; beechnuts are also a favorite food item (Elbroch & Rinehart, 2011), and many beech trees were located around this site as well. I also noted most gray squirrels detected at site #1 displayed nut collecting/burying behavior in the images collected.
Gray squirrel. Gray squirrels were detected, with no difference in abundance, at both sites.
Opossum. During the trapping period, opossum were detected, eight times, solely at the native site. The Student’s t-test revealed a statistical difference, (p=0.00, α=0.05), and supports the prediction for higher abundance of wildlife at the native site. Opossums are nomadic, usually not spending more than a few nights at any one area; they are omnivorous, with their favorite food preference being insects (Elbroch & Rinehart, 2011).
Brown rat. There was a statistically higher number of encounters of brown rat (Rattus norvegicus), at the Japanese knotweed site. Similarly, to the circumstances with the raccoons, one or two rats appeared to inhabit a burrow under the fallen log at site #2; images revealed the rat(s) entering under the logs at the same location at night. This may have similarly skewed this result.
Birds
While several species of birds were detected at both sites, there was no statistical difference between numbers of daily encounters of bird species at the two study sites (p=0.06, α=0.05). However, there was a decline shown in the variety of bird species, (decreased species richness) at the invasive site. Utilizing the species richness and abundance data gathered, the Shannon Wiener and Simpson diversity indices (Figure 2) were calculated. The Shannon Wiener diversity index was 1.75 for the native site, and 1.25 for the invasive site. The Simpson index was 4.28 at native site, compared to 1.61 at the invasive site. Both diversity indices showed a decline in the biodiversity of bird species at the site with Japanese knotweed. I think the data obtained from the camera traps were useful to investigate the effect of the presence of Japanese knotweed on wildlife abundance and diversity. The next step I look forward to investigating is comparing abundance and diversity of wildlife at a site(s) pre-removal and post-removal of Japanese knotweed (or other invasive vegetation). The direct comparison at the same site can diminish any variables that may have created biases in this project upon comparing two different locations.
Action and Reflection
Currently I am a zookeeper in the Bronx Zoo’s Mammal department. The Bronx Zoo is managed by the Wildlife Conservation Society (WCS). The WCS publishes an active online blog, “Wild View: An eye on wildlife”. Posts are typically a photo and a short write up pertaining to their mission to highlight modern conservation challenges from around the globe. Contributors range from in-situ WCS researchers in remote areas to zookeepers and volunteers. I wrote a post for the blog about my camera trap project and its results (Medina, 2016). The curator of the blog was excited to receive and share my post, as this piece fits into the mission of the blog, and discusses an important conservation topic of both local and global interest.
I plan to present my project and results to Bronx Zoo keepers, and others interested, via our local chapter of the American Association of ZooKeepers (AAZK) at a future meeting. I plan to use that future opportunity to boost interest in keepers assisting in a possible collaborative project with the Bronx River Alliance (BxRA), involving removal of invasive vegetation along the Bronx River on zoo grounds. I am in contact with the BxRA’s Ecology Director, Michelle Luebke. She has expressed interest in my project and collaborating on my future project idea. Linking invasive vegetation to wildlife may draw more attention to this important conservation issue. I would like to install camera traps at a site marked for future invasive vegetation removal. I think it would be valuable to obtain data on relative abundance and biodiversity pre- and post-removal of invasive plants for comparison. I plan to attend the Bronx River Watershed Summit on December 1, 2016, which seeks to foster citizen engagement and stewardship, to network, discuss and explore our possible collaboration and future opportunities. I am excited to develop this future project into my Master Plan.
I enjoyed working on this project. I learned more about camera traps and their applied use, especially how quickly overwhelmed you can become with the large amounts of data/images collected in a relatively short time. I was glad I used the settings that I did when deploying the cameras. While using the eight shot/multi-shot mode created many more images to process, in many instances if fewer images had been recorded per event, I would have missed the animal that triggered the camera. I was excited to actually collect potentially useful data. The data can be used to promote the importance of removal of invasive vegetation along the Bronx River and any area where invasive vegetation occurs. I am also looking forward to continuing to use camera traps and being surprised at what I may encounter. My favorite images collected during this project were of a young Cooper’s hawk.
I plan to present my project and results to Bronx Zoo keepers, and others interested, via our local chapter of the American Association of ZooKeepers (AAZK) at a future meeting. I plan to use that future opportunity to boost interest in keepers assisting in a possible collaborative project with the Bronx River Alliance (BxRA), involving removal of invasive vegetation along the Bronx River on zoo grounds. I am in contact with the BxRA’s Ecology Director, Michelle Luebke. She has expressed interest in my project and collaborating on my future project idea. Linking invasive vegetation to wildlife may draw more attention to this important conservation issue. I would like to install camera traps at a site marked for future invasive vegetation removal. I think it would be valuable to obtain data on relative abundance and biodiversity pre- and post-removal of invasive plants for comparison. I plan to attend the Bronx River Watershed Summit on December 1, 2016, which seeks to foster citizen engagement and stewardship, to network, discuss and explore our possible collaboration and future opportunities. I am excited to develop this future project into my Master Plan.
I enjoyed working on this project. I learned more about camera traps and their applied use, especially how quickly overwhelmed you can become with the large amounts of data/images collected in a relatively short time. I was glad I used the settings that I did when deploying the cameras. While using the eight shot/multi-shot mode created many more images to process, in many instances if fewer images had been recorded per event, I would have missed the animal that triggered the camera. I was excited to actually collect potentially useful data. The data can be used to promote the importance of removal of invasive vegetation along the Bronx River and any area where invasive vegetation occurs. I am also looking forward to continuing to use camera traps and being surprised at what I may encounter. My favorite images collected during this project were of a young Cooper’s hawk.
Conclusions
Two camera traps were simultaneously deployed, for 42 days, at two different sites to investigate if Japanese knotweed, a highly invasive plant, negatively affects wildlife along the Bronx River, Bronx, NY. One site had a large stand of Japanese knotweed present and the other did not have any knotweed immediately present in the area. The image data collected was utilized to measure species richness, relative abundance, and biodiversity to subsequently compare and determine if any differences were present. While Japanese knotweed did not appear to affect most mammals in a negative manner when reviewing encounter data, using the Shannon Wiener and Simpson diversity indices, the site with Japanese knotweed showed a decline in biodiversity of both mammals and birds.
I plan to utilize camera traps to further investigate and monitor possible changes in species richness, relative abundance and diversity at sites pre- and post-removal of Japanese knotweed. This future project can potentially yield useful data to draw attention to the important issue of invasive vegetation and its negative effects on wildlife communities and biodiversity.
I plan to utilize camera traps to further investigate and monitor possible changes in species richness, relative abundance and diversity at sites pre- and post-removal of Japanese knotweed. This future project can potentially yield useful data to draw attention to the important issue of invasive vegetation and its negative effects on wildlife communities and biodiversity.
Literature Cited
Bowkett, A., Rovero, F., & Marshall, A., (2007). The use of camera-trap data to model habitat use by antelope species in the Udzungwa Mountain forests, Tanzania. African Journal of Ecology, 45, 479-487.
Burton, A.C., Neilson, E., Moreira, D., Ladie, A., Steenweg, R., Fisher, J.T., Bayne, & Boutin, S. (2015). Wildlife camera trapping: a review and recommendations for linking surveys to ecological processes. Journal of Applied Ecology, 52, 675-685.
Christensen, D.R. (2016). A simple approach to collecting useful wildlife data using remote camera-traps in undergraduate biology courses. Bioscene, 42(1), 25-31.
Elbroch, M., & Rinehart, K. (2011). Peterson reference guide to behavior of North American mammals. New York, NY: Houghton Mifflin Harcourt Publishing Company.
Gerber, E., Krebs, C., Murrell, C., Moretti, M., Rocklin, R., & Schaffner, U. (2008). Exotic invasive knotweeds (Fallopia spp.) negatively affect native plant and invertebrate assemblages in European riparian habitats. Biological Conservation, 141, 646-654.
Haight, C.A., Schular, J.A., McCarthy, K., Tobing, S.L., Kreisberg, R., Larson, M., & Palmer, M. (2014, October). Japanese knotweed management in the riparian zone of the Bronx River: The effect of different removal techniques on sapling growth and the herbaceous layer. Poster presented at Reclaiming a river: Conservation & Community Symposium, WCS/Bronx Zoo.
Hamel, S., Killengreen, S.T., Henden, J., Roed-Eriksen, L., Ims, R., & Yoccoz, N.G. (2013). Towards good practice guidance in using camera-traps in ecology: influence of sampling design on validity of ecological inferences. Methods in Ecology and Evolution, 4, 105-113.
Karlin, M. & De La Paz, G. (2015). Using camera-trap technology to improve undergraduate education and citizen-science contributions in wildlife research. The Southwestern Naturalist, 60(2-3), 171-179.
Maerz, J.C., Blossey, B., & Nuzzo, V. (2005). Green frogs show reduced foraging success in habitats invaded by Japanese knotweed. Biodiversity and Conservation, 14, 2901-2911.
Medina, M. (2014). Restoration projects and invasive Japanese knotweed (Fallopia japonica) along the Bronx River, New York City. Community Engagement Lab. WBLC Conservation Science and Community 18
Medina, M. (2016, December,1). Wildlife vs. plant invaders. Retrieved from http://blog.wcs.org/photo/2016/12/01/wildlife-vs-plant-invaders-bronx-river-hawk/
Newey, S., Davidson, P., Nazir, S., Fairhurst, G., Verdicchio, F., Irvine, R.J., & van der Wal, R. (2015). Limitations of recreational camera traps for wildlife management and conservation research: a practitioner’s perspective. AMBIO: A Journal of the Human Environment, 44(4), S624- S635.
O’Brien, T.G. (2010). Wildlife picture index and biodiversity monitoring: issues and future directions. Animal Conservation, 13, 350-352.
O’Brien, T.G. (2011). Abundance, density and relative abundance: a conceptual framework. In A.F. O’Connell, J.D. Nichols & K.U. Karanth (Eds.), Camera Traps in Animal Ecology (pp71-73). New York, NY: Springer.
Rowcliffe, J.M., Field, J., Turvey, S.T., & Carbone, C. (2008). Estimating animal density using camera traps without the need for individual recognition. Journal of Applied Ecology, 45,1228-1236.
Swann, D.E., Hass, C.C., Dalton, D.C., & Wolf, S.A. (2004). Infrared-triggered cameras for detecting wildlife: an evaluation and review. Wildlife Society Bulletin, 32(2). 357-365.
Trolliet, F., Huynen, M., Vermeulen, C., & Hambuckers, A. (2014). Use of camera traps for wildlife studies. A review. Biotechnologie Agronomie Societe et Environment,18(3); 446-454.
United States Department of Agriculture (USDA)-Forest Health Technology Enterprise Team. (2016). Japanese knotweed biological control. [Fact sheet]. Retrieved from http://www.fs.fed.us/foresthealth/technology/pdfs/FS_jaknotweed.pdf
Urgenson, L.S., Reichard, S.H., & Halpern, C.B. (2009). Community and ecosystem consequences of giant knotweed (Polygonum sachalinense) invasion into riparian forests of western Washington, USA. Biological Conservation, 142, 1536-1541.
Weston, L.A., Barney, J.N., & DiTommaso, A. (2005). A review of the biology and ecology of three invasive perennials in New York State: Japanese knotweed (Polygonum cuspidatum), mugwort (Artemisia vulgaris) and pale swallow-wort (Vincetoxicum rossicum). Plant and Soil, 277, 53-69.
Burton, A.C., Neilson, E., Moreira, D., Ladie, A., Steenweg, R., Fisher, J.T., Bayne, & Boutin, S. (2015). Wildlife camera trapping: a review and recommendations for linking surveys to ecological processes. Journal of Applied Ecology, 52, 675-685.
Christensen, D.R. (2016). A simple approach to collecting useful wildlife data using remote camera-traps in undergraduate biology courses. Bioscene, 42(1), 25-31.
Elbroch, M., & Rinehart, K. (2011). Peterson reference guide to behavior of North American mammals. New York, NY: Houghton Mifflin Harcourt Publishing Company.
Gerber, E., Krebs, C., Murrell, C., Moretti, M., Rocklin, R., & Schaffner, U. (2008). Exotic invasive knotweeds (Fallopia spp.) negatively affect native plant and invertebrate assemblages in European riparian habitats. Biological Conservation, 141, 646-654.
Haight, C.A., Schular, J.A., McCarthy, K., Tobing, S.L., Kreisberg, R., Larson, M., & Palmer, M. (2014, October). Japanese knotweed management in the riparian zone of the Bronx River: The effect of different removal techniques on sapling growth and the herbaceous layer. Poster presented at Reclaiming a river: Conservation & Community Symposium, WCS/Bronx Zoo.
Hamel, S., Killengreen, S.T., Henden, J., Roed-Eriksen, L., Ims, R., & Yoccoz, N.G. (2013). Towards good practice guidance in using camera-traps in ecology: influence of sampling design on validity of ecological inferences. Methods in Ecology and Evolution, 4, 105-113.
Karlin, M. & De La Paz, G. (2015). Using camera-trap technology to improve undergraduate education and citizen-science contributions in wildlife research. The Southwestern Naturalist, 60(2-3), 171-179.
Maerz, J.C., Blossey, B., & Nuzzo, V. (2005). Green frogs show reduced foraging success in habitats invaded by Japanese knotweed. Biodiversity and Conservation, 14, 2901-2911.
Medina, M. (2014). Restoration projects and invasive Japanese knotweed (Fallopia japonica) along the Bronx River, New York City. Community Engagement Lab. WBLC Conservation Science and Community 18
Medina, M. (2016, December,1). Wildlife vs. plant invaders. Retrieved from http://blog.wcs.org/photo/2016/12/01/wildlife-vs-plant-invaders-bronx-river-hawk/
Newey, S., Davidson, P., Nazir, S., Fairhurst, G., Verdicchio, F., Irvine, R.J., & van der Wal, R. (2015). Limitations of recreational camera traps for wildlife management and conservation research: a practitioner’s perspective. AMBIO: A Journal of the Human Environment, 44(4), S624- S635.
O’Brien, T.G. (2010). Wildlife picture index and biodiversity monitoring: issues and future directions. Animal Conservation, 13, 350-352.
O’Brien, T.G. (2011). Abundance, density and relative abundance: a conceptual framework. In A.F. O’Connell, J.D. Nichols & K.U. Karanth (Eds.), Camera Traps in Animal Ecology (pp71-73). New York, NY: Springer.
Rowcliffe, J.M., Field, J., Turvey, S.T., & Carbone, C. (2008). Estimating animal density using camera traps without the need for individual recognition. Journal of Applied Ecology, 45,1228-1236.
Swann, D.E., Hass, C.C., Dalton, D.C., & Wolf, S.A. (2004). Infrared-triggered cameras for detecting wildlife: an evaluation and review. Wildlife Society Bulletin, 32(2). 357-365.
Trolliet, F., Huynen, M., Vermeulen, C., & Hambuckers, A. (2014). Use of camera traps for wildlife studies. A review. Biotechnologie Agronomie Societe et Environment,18(3); 446-454.
United States Department of Agriculture (USDA)-Forest Health Technology Enterprise Team. (2016). Japanese knotweed biological control. [Fact sheet]. Retrieved from http://www.fs.fed.us/foresthealth/technology/pdfs/FS_jaknotweed.pdf
Urgenson, L.S., Reichard, S.H., & Halpern, C.B. (2009). Community and ecosystem consequences of giant knotweed (Polygonum sachalinense) invasion into riparian forests of western Washington, USA. Biological Conservation, 142, 1536-1541.
Weston, L.A., Barney, J.N., & DiTommaso, A. (2005). A review of the biology and ecology of three invasive perennials in New York State: Japanese knotweed (Polygonum cuspidatum), mugwort (Artemisia vulgaris) and pale swallow-wort (Vincetoxicum rossicum). Plant and Soil, 277, 53-69.
Appendix